Select a Size
About This Item
Quality Level
assay
97%
form
solid
reaction suitability
reaction type: click chemistry
mp
109-111 °C
storage temp.
−20°C
SMILES string
COC(=O)c1ccc(cc1P(c2ccccc2)c3ccccc3)C(=O)Oc4c(F)c(F)c(F)c(F)c4F
InChI
1S/C27H16F5O4P/c1-35-27(34)18-13-12-15(26(33)36-25-23(31)21(29)20(28)22(30)24(25)32)14-19(18)37(16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-14H,1H3
InChI key
OURNVXDJALDDIG-UHFFFAOYSA-N
Application
- Staudinger ligation reagent for the conjugation of amine and azide containing compounds or biomolecules.
- The amine functionalized molecule first reacts with the phosphine through the activated pentafluorophenyl ester.The azide-molecule is then reacted with the newly labeled phosphine to form the iminophosphorane, and the aza-ylide is subsequently captured by the methyl ester to yield the covalent conjugated product.
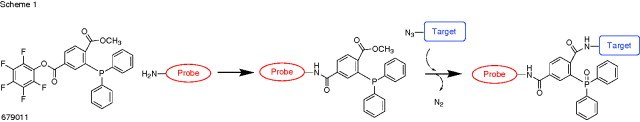
The Staudinger Ligation: A High-Yield, Chemoselective, and Mild Synthetic Method
Storage Class
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
The reaction between an azide and a phosphine forming an aza-ylide was discovered almost a century ago by Nobel Prize laureate Herrmann Staudinger.
Chemoselective ligation strategies are a key success factor for chemical biology research. Ligation techniques open pathways to fully synthetic large peptides and even proteins.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 679011-100MG | 04061833564882 |
| 679011-25MG | 04061833564899 |